
ITPC'S YEAR IN NUMBERS

Directly strengthened the advocacy capacity and HIV treatment knowledge of 392 activists across the globe; and these treatment activists went on to mobilize peers and galvanize policy makers to improve access to optimal treatment, including removing intellectual property barriers, promoting routine viral load testing, PrEP and differentiated service delivery.

Conducted 18 national and regional trainings to empower communities to undertake research, in support of generating evidence to support better advocacy outcomes and strategies.

Achieved approximately US$472 million in annualized cost savings for our intellectual property related interventions on key ARVs across the four focus countries of our Make Medicines Affordable campaign.

Established 12 community treatment observatories (CTOs) to systematically collect data, analyze & monitor trends along the HIV continuum cascade.

Launched 2 regional community advisory boards in Latin America and Caribbean and the Middle East and North Africa, two regions neglected by Pharma industry.

Supported 17 community-based organizations and civil society networks in three regions to take up national advocacy strategies to address critical treatment access issues through small grants.
CAMPAIGNS & INNOVATION
Calling Out Big Pharma & Their Patent Monopolies
Our #MakeMedicinesAffordable campaign is showing the lengths that Big Pharma will go to protect unmerited monopolies, keeping life saving drugs out of reach of those who need them most.

New Toolkit & Resources to Support Communities to Demand PrEP
People who need PrEP have a right to access it as a human right to health. Our new toolkit ensures communities have the knowledge they need to demand it.

New Toolkit Supports Communities to Demand Services That Work for Them
Communities want and need more effective ways of access the treatment they need. Our toolkit helps them engage in and demand recipient-centered models of care.
Community Monitoring of HIV Services in West Africa
Our #WatchWhatMatters initiative is increasing domestic oversight of HIV services among key populations in 11 countries in West Africa by supporting civil society to act as watchdogs.
REACH & RESULTS
With our regions, networks and partners, we ran projects to improve access to optimal HIV treatment in these countries in 2017:
- Groundwork laid for expanded community monitoring
- Training delivered on intellectual property and access to medicines
- Treatment activists mobilized to demand routine viral load testing
- Community-based research conducted
- Patent oppositions filed
- Treatment activists capacitated to participate in regional Community Advisory Boards
- Training delivered to increase capacity on HIV treatment, rights and advocacy
- IP intervention resulted in price reduction of 50% or more on at least two ARVs
- Treatment activists mobilized to demand differentiated service delivery
- Community monitoring systems established

- Groundwork laid for expanded community monitoring
- Zimbabwe
- Training delivered on intellectual property and access to medicines
- Algeria, Argentina, Armenia, Azerbaijan, Belarus, Benin, Bolivia, Burkina-Faso, Cameroon, Chile, Colombia, Congo, Costa Rica, Côte d’Ivoire, Dominican Republic, El Salvador, France, Georgia, Guatemala, Kazakhstan, Kyrgyzstan, Latvia, Lebanon, Mexico, Moldova, Morocco, Peru, Russia, Switzerland, Tajikistan, Tunisia, Ukraine, Uzbekistan, Venezuela
- Treatment activists mobilized to demand routine viral load testing
- India, Kenya, Malawi, South Africa, Tanzania, Thailand, Vietnam, Zimbabwe
- Community-based research conducted
- Morocco, Pakistan, Tunisia
- Treatment activists capacitated to participate in regional Community Advisory Boards
- Algeria, Armenia, Azerbaijan, Belarus, Bolivia, Chile, Costa Rica, Dominican Republic, Ecuador, Egypt, El Salvador, Georgia, Guatemala, Jordan, Kazakhstan, Kyrgyzstan, Latvia, Lebanon, Mauritania, Mexico, Morocco, Pakistan, Peru, Russia, Switzerland, Tajikistan, Tunisia, Ukraine, Uzbekistan, Venezuela
- Training delivered to increase capacity on HIV treatment, rights and advocacy
- Benin, Cote d’Ivoire, Gambia, Georgia, Ghana, Guinea Bissau, Guinea Conakry, India, Indonesia, Kenya, Kyrgyzstan, Liberia, Lithania, Malaysia, Mali, Mexico, Myanmar, Nepal, Senegal, Sierra Leone, Swaziland, Tajikistan, Tanzania, Thailand, Uganda, Ukraine, Vietnam, Zimbabwe
- IP intervention resulted in price reduction of 50% or more on at least two ARVs
- Brazil, Argentina, Thailand, Ukraine
- Patent oppositions filed
- Brazil, Ukraine
- Community monitoring systems established
- Benin, Côte d’Ivoire, The Gambia, Ghana, Guinea Bissau, Guinea Conakry, Kyrgyzstan, Liberia, Mali, Pakistan, Russia, Senegal, Sierra Leone, Togo, Ukraine
- Treatment activists mobilized to demand differentiated service delivery
- India, Kenya, Malawi, South Africa, Tanzania, Vietnam, Zimbabwe
SIGNIFICANT SAVINGS FOR HEALTH BUDGETS
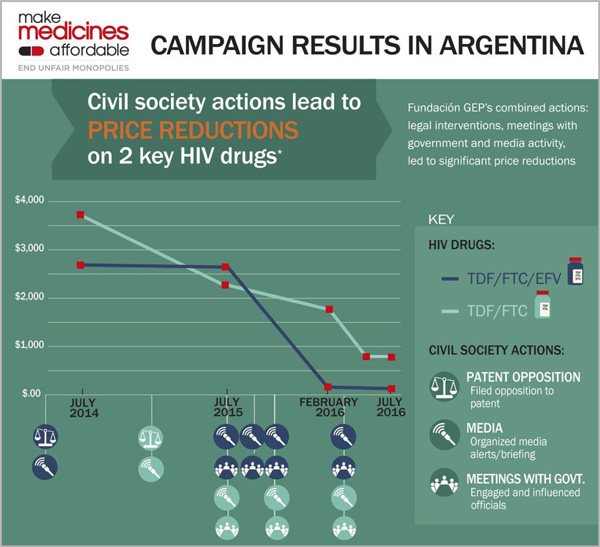
With our consortium partners, our work to make medicines affordable in four focus countries contributed to projected, combined savings of US$472 million per year.
In Argentina, US$50 million annual savings on two HIV drugs, means 27,000 more people could receive HIV treatment for 10 years.
See our full infographic about the impact of our interventions in Argentina.
GLOBAL INFLUENCE & VISIBILITY
In 2017, ITPC participated in meetings and conferences to present community-based research, influence international policy and amplify the voices of the people we represent.

“When you meaningfully invest in communities, you can do more and you can do better!”
ITPC Executive Director Solange Baptiste spoke at the IAS 2017 Official Press Event on Impacts of Investing in HIV highlighting the unintended consequences of the shift to domestic funding and why community investment and strengthening is critical.

“Gilead Sciences is funding this conference and making a lot of positive public relations, but we all know that its pricing practices leave millions of people without access to lifesaving treatment!”
ITPC IP and Access to Medicines Lead Othoman Mellouk delivered the community address at the closing plenary.

ITPC MENA led activists in action against ViiV Healthcare, whose voluntary license for dolutegravir (DTG) includes all countries in Africa except Algeria.
The actions and activities gained significant visibility far beyond the conference itself, with social media coverage reaching more than 27,000 impressions (persons reached) each day.
You can see the highlights from each day of the conference here
VALUE FOR MONEY
ITPC continued to represent excellent value for money in 2017.
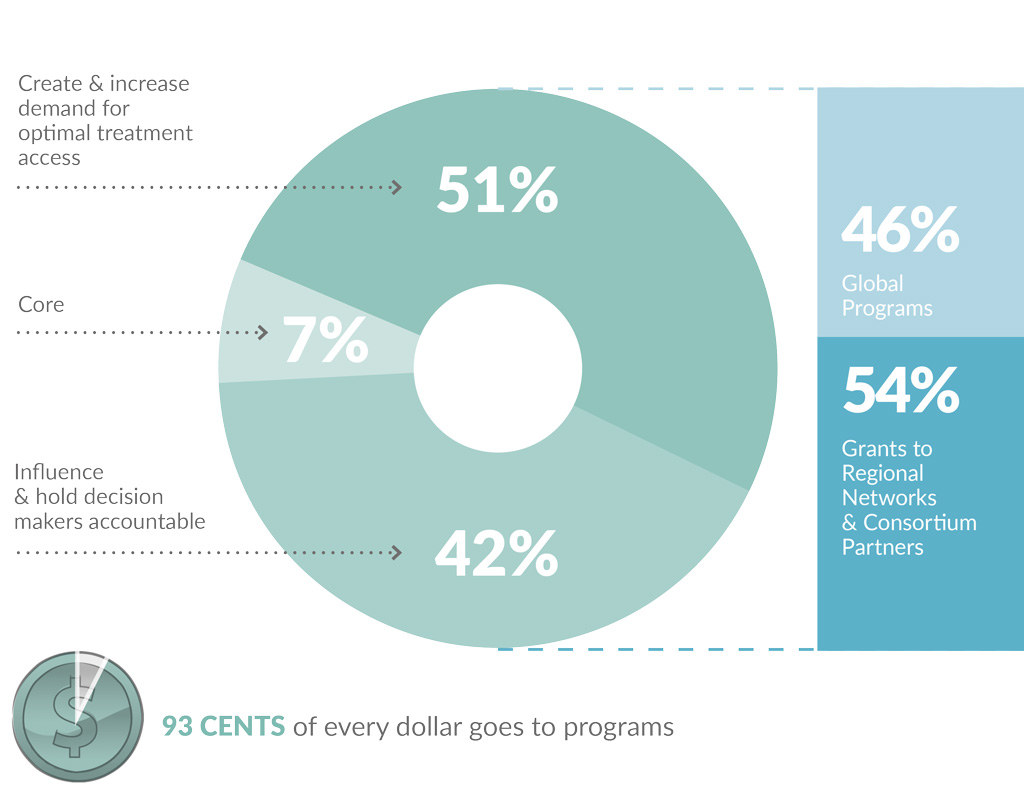
Just 7 cents in every dollar goes to administration, finance, development and communications.
HIGHLIGHTS OF THE YEAR 2017

JANUARY
The Global Fund gives ITPC final approval to support the Regional Community Treatment Observatory in West Africa (RCTO-WA). This decision by the Global Fund was eagerly welcomed by communities in West Africa who are facing unacceptable challenges getting access to HIV treatment.
FEBRUARY
ITPC and ITPC West Africa officially launch the RCTO-WA, extending community based monitoring of HIV treatment access to 11 West African countries.
See highlights from the launch event in Abidjan
MARCH
ITPCru publishes extensive report on the analysis of 4500 public procurement monitoring data for ARVs in Russia. The report puts forth recommendations for improving drug provision in the country.
ITPC South Asia expresses outraged following a decision by the Indian Parliament to pass the HIV/AIDS Prevention & Control Bill and absolving itself from responsibility of providing treatment for people living with HIV in India.
Advocates, activists, and researchers took to ITPC’s listserv to share strong reactions and spark discussion on the development of WHO guidelines on the management of advanced HIV disease (including same day start).
APRIL
The RCTO-WA builds critical connections at regional level, positioning the project as a key player in the UNAIDS Catch-Up Plan.
MAY
ITPC kicks off a consultative process on PrEP by hosting the Communityled Consultative Think Tank Meeting on PrEP Access and Use in Port of Spain, Trinidad & Tobago. The meeting informed the development of a global position statement and key population activist toolkit on PrEP.
Read the full meeting report here
Bridging the Gaps Alliance partners draft declaration on the right of people who use drugs to be protected from state sanctioned violence, following extrajudicial killings in the Philippines and elsewhere.
JUNE
ITPC and the AIDS and Rights Alliance of Southern Africa (ARASA) host a regional workshop on differentiated service delivery for activists from Asia and Southern Africa.
The RCTO-WA conducts a baseline assessment, rolls out a digital project database and archiving system, and continues to build capacity for community monitoring through a series of intense trainings.
JULY
ITPC LATCA and allies urge Guatemala and the international community to expand HIV treatment coverage through the use of compulsory licensing for fixed-dose combination tablet.
ITPC South Asia condemns the harmful provisions included in the Regional Comprehensive Economic Partnership (RCEP), which undermine access to affordable medicines across the Asia Pacific region.
At the IAS conference in Paris, ITPC participates in numerous sessions to ensure visibility and action on HIV treatment acesss. At the closing plenary, ITPC IP and Access to Medicines Lead Othoman Mellouk delivers the community address.
Read a summary of our activities
ITPC MENA celebrates a victory as Bristol-Myers Squibb extends voluntary license on atazanavir to Egypt.
AUGUST
On International Youth Day, ITPC reflects on the future of the HIV response through the eyes of youth leadership.
Read a blog post about the Intergenerational Summit on Positive Youth Leadership
SEPTEMBER
Director of Global Programs and Advocacy Wame Mosime speaks to donors about civil society driven actions on sustainability and domestic mobilization for HIV at the FCAA Philantrophy Summit in Washington D.C.
Read a summary of the session here
OCTOBER
IP and Access to Medicines Lead Othoman Mellouk addresses the Human Rights Council’s 2017 Social Forum in Geneva, Switzerland explaining why we can’t stand back while Big Pharma abuses the patent system at the expense of public health
ITPC MENA releases comparative study on intellectual property provision in Tunisia, Morocco, and Egypt. The study assesses intellectual property provisions in different bodies of laws that have an impact on access to medicines in the three countries.
Read about the study here available in English, French and Arabic
NOVEMBER
Prevention Access Campaign and ITPC announce a groundbreaking partnership to amplify the Undetectable=Untransmittable (U=U) message among communities of people living with HIV worldwide.
Read the statement about the partnership
ITPC and ARASA, in collaboration with the IAS, develop a new toolkit – What Works for Me: Activist Toolkit on Differentiated Service Delivery – to empower people living with HIV to access treatment options that best meet their needs.
Read about and download the toolkit
DECEMBER
At ICASA 2017 in Abidjan, Cote d’Ivoire, ITPC leads, co-hosts and participates in sessions to raise the flag for optimal HIV treatment. Every day, ITPC leads satellite sessions, protest actions against pharmaceutical companies, and hosts the Treatment Networking Zone.
Read a summary of our activities
ITPC publishes the Community Demand Creation Model in a special supplement of the Journal of the International AIDS Society (JIAS).
Consortium partner ABIA, challenge ViiV Healthcare’s patent application on key HIV drug, dolutegravir (DTG) in Brazil. Brazil currently procures DTG at a cost of US$ 547.50 per patient per year, if a patent monopoly is ruled out then generic versions could be sourced for around $44 per patient per year.
THANK YOU
OUR WORK WOULDN’T BE POSSIBLE WITHOUT YOU.
THANK YOU FOR ALL YOUR SUPPORT IN 2015.
